Extracellular vesicles (EVs) are nanometer-scale, lipid bilayer enclosed structures that are secreted by cells. EVs elicit complex signaling functions in other cells that encounter these vesicles. EVs can be found in biological fluids and media of cultured cells, and exosomes are a subset of EVs. While EVs or exosomes for therapeutic use immediately conjure thoughts of mesenchymal stem/stromal cells, exosomes are secreted by many cell types, including immune cells. In addition to being active players in cell-cell interactions and waste elimination, immune cell-derived exosomes play an important role in antigen presentation and antitumor responses.1 Therefore, exosomes derived from immune cells are attractive candidates for clinical applications ranging from diagnostics to therapeutics.
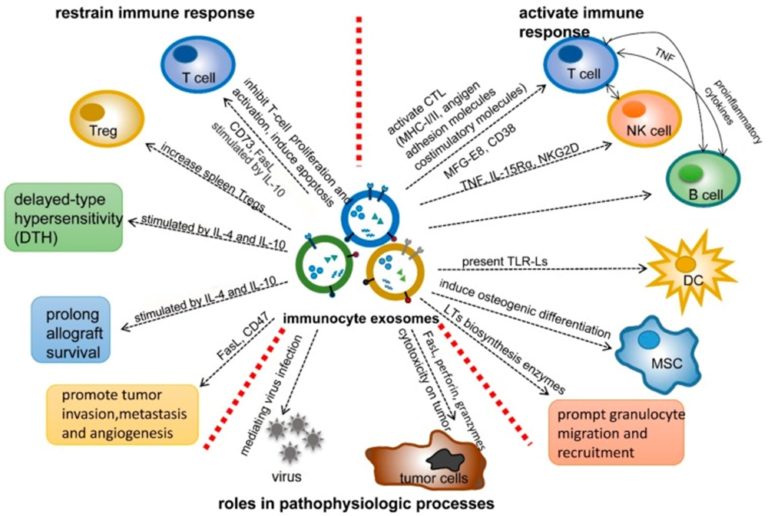
Natural Killer (NK) cell-derived exosomes contain a variety of cytotoxic molecules, including perforin, granzymes A and B granulysin, and Fas ligand. Following exposure to neuroblastoma cells, NK activation resulted in increased exosomes secretion compared to unstimulated NK cells.1 Additionally, exosomes from NK cells displayed anti-melanoma activity both in vitro and in vivo.2 In a study by Wu et al., NK-derived exosomes were isolated in large quantities following activation by different tumor cell lines.3 Although the various killer exosome proteins exhibited different mechanisms of action, each of the molecules contributed equally to the activity against the various target cells.3
Unlike NK cells, macrophages and dendritic cells (DCs) secrete exosomes continuously. Macrophages produce chemokines which recruit lymphocytes and induce an inflammatory reaction.4 In contrast, DCs are able to induce more specific immune responses. DC-derived exosomes, also called dexosomes, are involved in antigen-presentation in the context of MHC (Major Histocompatibility Complex) along with co-stimulatory molecules which drive antigen-specific T cell proliferation.1 Dexosomes are also capable of directly binding NK cells for activation and release of cytotoxic proteins and apoptosis-inducing molecules.5 Clinical studies of dexosomes have been utilized to treat metastatic melanoma, colorectal cancer, and non-small cell lung carcinoma.6
Lastly, as opposed to stimulatory exosomes, regulatory T (Treg) cell-derived exosomes have been used to suppress immune response. In a kidney transplantation rat model, Treg-derived exosomes were shown to suppress T cell proliferation resulting in delayed graft rejection.7 Treg-derived exosomes may also be used to alleviate chronic inflammation as well as to treat autoimmune diseases.1
Exosomes have a natural feature which allows for delivery of tumor antigens or therapeutic molecules in a cell-free approach. Understanding how to best exploit the clinical benefit of exosomes is still underway. Researchers have been focused on optimization of large scale and consistent production and isolation of exosomes for clinical use. Engineering exosomes for therapies has also been on the rise and can present potential treatments against a variety of diseases and conditions.8 The lack of toxicity and immunogenicity allows for safe and repeated administration of exosomes and makes exosomes a promising candidate for immunotherapy. Exosomes from cord blood or adult peripheral blood pre-isolated immune cells from OrganaBio can provide excellent source material for your studies and accelerate translation into the clinic.
References
- Li Q, Wang H, Peng H, Huyan T, Cacalano NA. Exosomes: Versatile Nano Mediators of Immune Regulation. Cancers (Basel). 2019;11(10):1557. Published 2019 Oct 14. doi:10.3390/cancers11101557
- Zhu L, Kalimuthu S, Gangadaran P, et al. Exosomes Derived From Natural Killer Cells Exert Therapeutic Effect in Melanoma. Theranostics. 2017;7(10):2732–2745. Published 2017 Jul 7. doi:10.7150/thno.18752
- Wu CH, Li J, Li L, et al. Extracellular vesicles derived from natural killer cells use multiple cytotoxic proteins and killing mechanisms to target cancer cells. J Extracell Vesicles. 2019;8(1):1588538. Published 2019 Mar 12. doi:10.1080/20013078.2019.1588538
- McDonald MK, Tian Y, Qureshi RA, et al. Functional significance of macrophage-derived exosomes in inflammation and pain. Pain. 2014;155(8):1527–1539.
- Wen C, Seeger RC, Fabbri M, Wang L, Wayne AS, Jong AY. Biological roles and potential applications of immune cell-derived extracellular vesicles. J Extracell Vesicles. 2017;6(1):1400370. Published 2017 Nov 22. doi:10.1080/20013078.2017.1400370
- Samuel M, Gabrielsson S. Personalized medicine and back-allogeneic exosomes for cancer immunotherapy [published online ahead of print, 2019 Jul 29]. J Intern Med. 2019;10.1111/joim.12963. doi:10.1111/joim.12963
- Yu X, Huang C, Song B, et al. CD4+CD25+ regulatory T cells-derived exosomes prolonged kidney allograft survival in a rat model. Cell Immunol. 2013;285(1-2):62–68. doi:10.1016/j.cellimm.2013.06.010
- Mendt M, Kamerkar S, Sugimoto H, et al. Generation and testing of clinical-grade exosomes for pancreatic cancer. JCI Insight. 2018;3(8):e99263. Published 2018 Apr 19. doi:10.1172/jci.insight.99263






