Breakthroughs in immunotherapy, such as chimeric antigen receptor T cell (CAR-T) therapy and checkpoint inhibition antibodies, have led to a rejuvenated interest in the human immune system. Insights from basic immunology research drive approaches and innovations in the exploding immunotherapy field (Figure 1). Studies of tumor-infiltrating lymphocytes helped shed light on the complex tumor microenvironment and the mechanisms of cancer immune evasion. Discoveries of neo-antigens, unique biomarkers, and optimization of combination immunotherapy (Figure 1) have led to improved efficacy and patient outcomes. However, we still don’t fully understand the complex relationship between immunity and disease, especially when it comes to cell activation, cytotoxicity, regulation, and memory.
Immune cells are the cellular pillars of immunotherapy, and therefore, understanding immune responses is the key to developing new therapeutic strategies for cancer and infectious disease treatment. Tightly regulated surveillance by immune cells can distinguish between healthy, normal cells and unhealthy, abnormal cells, such as pathogen-infected, tumor, or auto-reactive cells. Effective immune responses could either eradicate infected and tumor cells or impair their phenotype and function.
Each of the various immune cell types has a unique role. Lymphocytes are part of the adaptive immune system, which include B cells and T cells. B cells play a major role in antibody-mediated immune responses, whereas T cells are involved in cell-mediated killing. Due to the ability of T cells to specifically recognize foreign antigens via the T cell receptor, various T cell products have been engineered to efficiently recognize viral infected cells and cancer cells. A couple of examples are Yescarta® (by Kite, a Gilead company) and Kymriah® (by Novartis), which were pioneers in FDA approval of Chimeric Antigen Receptor T cell (CAR-T cell) therapy. Additionally, the immunomodulatory properties of T regulatory cells have been utilized in organ transplantation and in the prevention and treatment of graft-versus-host disease and autoimmunity.
Innate immune cells, composed of natural killer (NK) cells, eosinophils, basophils, neutrophils, monocytes, macrophages, and dendritic cells, participate in initial immune responses by direct attack or indirect triggering of adaptive immune cell activation. In contrast to antigen-dependent T cells, NK cells are potent cytotoxic immune cells which exhibit a lower threshold of activation compared with other lymphocytes. Being ‘first-responders’ in viral infections and their unique ability to form memory, make NK cells ideal “off-the-shelf” allogeneic cell therapy candidates. Whether primary or genetically-modified, NK cells are emerging as major players in immunotherapy development.
By now, it has been established how important immunotherapy is for the treatment of cancer and other diseases. Personalized treatment approaches and combination therapy have shown great benefits to patients and improved survival. However, therapeutic optimization of multi-agent regimens is the most challenging. Therefore, it is vital for researchers and clinicians to understand the nature of our bodies’ immunobiology. While extremely complicated, looking ahead, we anticipate the advancement of immunotherapy to continue with the rapid growth of knowledge. In addition, with the successful application of autologous immunotherapies, such as Yescarta and Kymriah, the industry is now turning its eye to the development of allogeneic immunotherapies to treat many more patients, and at a more affordable price point. At OrganaBio, we’re working diligently to proactively address bottlenecks in allogeneic immunotherapy and to develop solutions that will enable researchers to rapidly move these therapies to clinic.
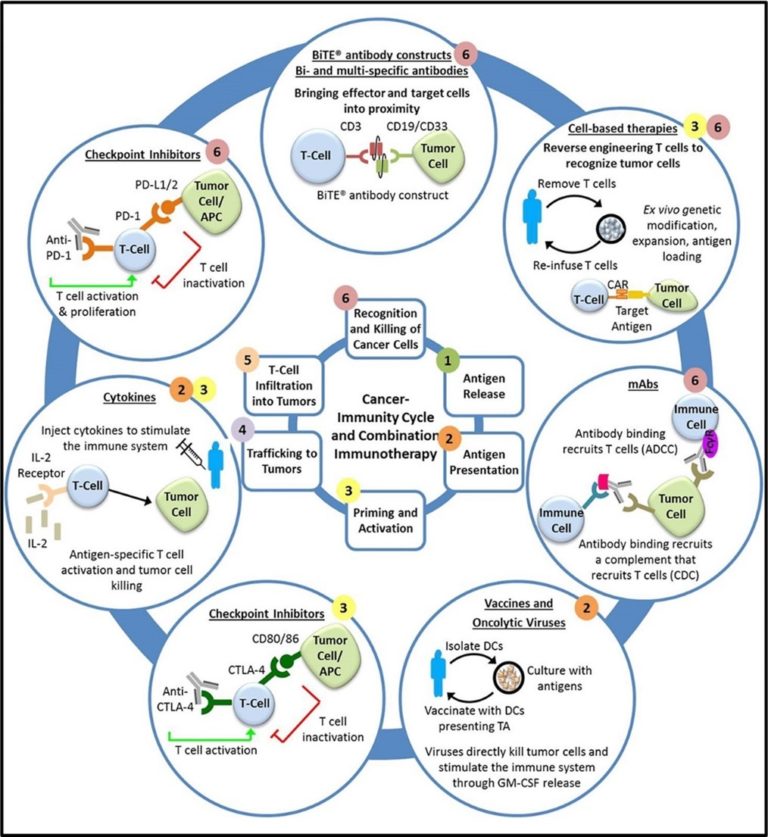
Figure 1 The Cancer Immunity Cycle and Combination Immunotherapy. The numbers represent the phases of disease in which the immunotherapy reagent acts either alone or in combination with other agents. Morrissey, K., Yuraszeck, T., Li, C.‐C., Zhang, Y. and Kasichayanula, S. (2016), Immunotherapy and Novel Combinations in Oncology: Current Landscape, Challenges, and Opportunities. Clinical And Translational Science, 9: 89-104.






